Certificate of Analysis
Lab reports coming soon. HPLC, Mass Spec & endotoxin testing.
MSDS
Material Safety Data Sheet coming soon. Handling & storage info for laboratory use.
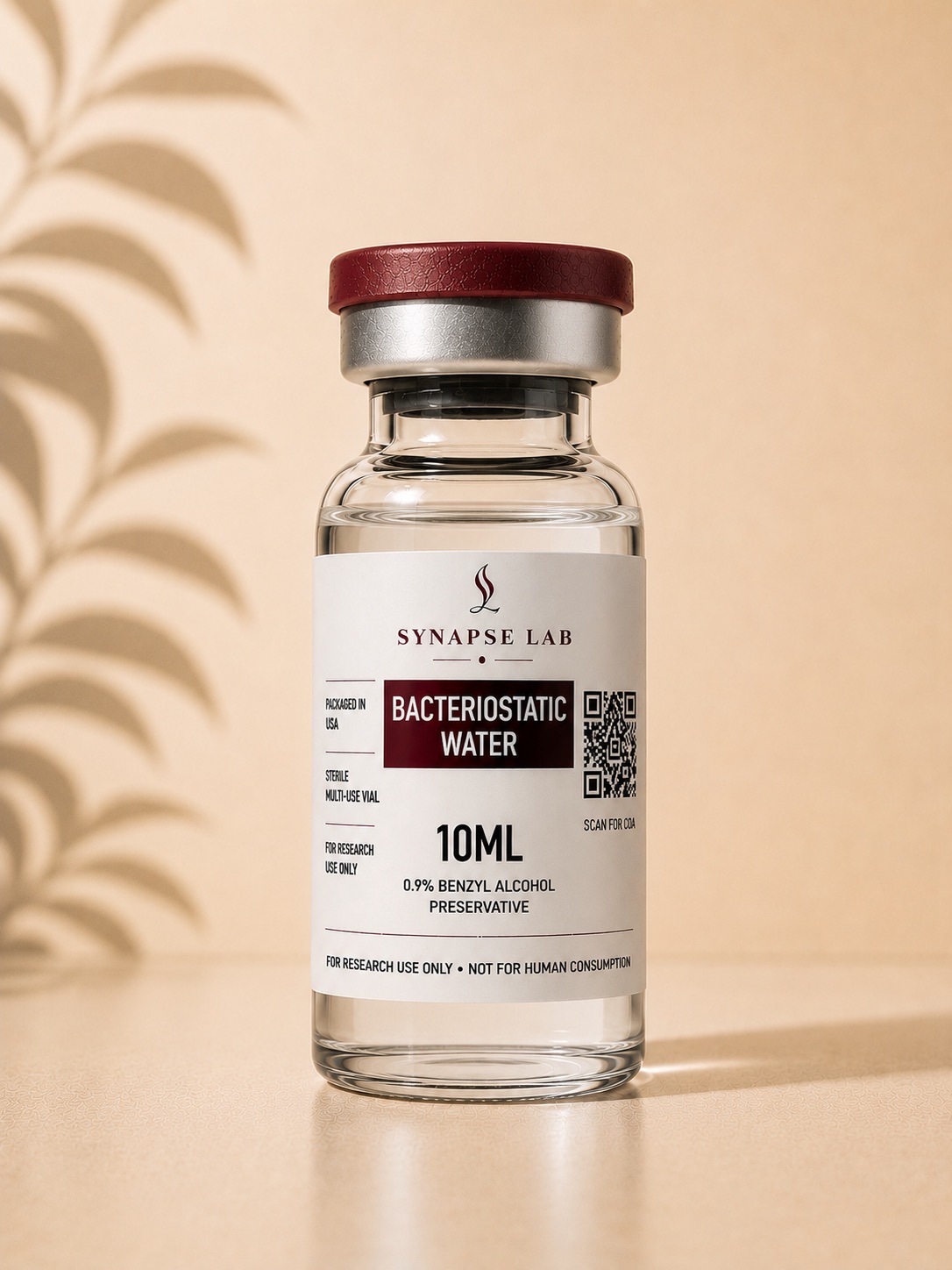
Bacteriostatic Water
Research Supply
Compound Overview
Bacteriostatic Water (BAC Water) is a sterile, non-pyrogenic preparation of water containing 0.9% benzyl alcohol as a bacteriostatic preservative. Within laboratory and peptide research environments, BAC Water is commonly utilized as a diluent for the reconstitution of lyophilised compounds and research peptides prior to experimental handling and analytical use. The inclusion of benzyl alcohol is intended to inhibit the growth of certain microorganisms following initial vial access, supporting short-term multi-use handling within controlled laboratory settings. Researchers commonly utilize BAC Water during peptide solubility studies, concentration preparation protocols, volumetric analysis, and compound reconstitution procedures. BAC Water is not considered an active research compound itself, but rather a laboratory-grade auxiliary preparation designed to support the accurate handling and preparation of lyophilised materials used within scientific and analytical research environments. Manufactured under sterile processing standards, each batch undergoes independent third-party analytical verification to support sterility, identity, consistency, and batch traceability standards across all Synapse Lab products.
- Independently tested through ISO 17025 accredited laboratories
- Sterility and endotoxin verification performed on every batch
- Identity and composition confirmation
- Full-panel third-party analytical testing
- QR-coded batch traceability and Certificate of Analysis (COA) access provided
- Batch-specific laboratory verification for consistency and quality assurance
Product Type: Sterile Bacteriostatic Water Composition: Sterile Water with 0.9% Benzyl Alcohol Appearance: Clear sterile liquid Application: Peptide and lyophilised compound reconstitution Pack Size: 10ml Multi-Use Vial
- Reconstitution of lyophilised research peptides
- Laboratory dilution and volumetric preparation
- Peptide concentration adjustment protocols
- Analytical and biochemical research handling procedures
- Scientific research and development environments
- Store unopened vial at controlled room temperature
- Following initial use, refrigeration at 2–8°C is recommended to help preserve sterility
- Protect from excessive heat, freezing, and direct light exposure
- Maintain sterile handling procedures during use
- Intended for laboratory handling and peptide reconstitution procedures only
- Use appropriate sterile laboratory technique during vial access and transfer
- Discontinue use if solution becomes cloudy or particulate matter is observed
- Avoid unnecessary repeated environmental exposure following opening
This product is intended strictly for scientific research and laboratory applications only. Not intended for diagnostic, therapeutic, cosmetic, veterinary, or medical use.
The Synapse Quality Standard
Every compound undergoes a rigorous multi-stage verification process before receiving our seal of approval.
HPLC Testing
High-performance liquid chromatography ensures precise purity measurement for every synthesized batch.
Mass Spec Verification
Mass spectrometry confirms molecular identity and detects trace impurities at parts-per-billion sensitivity.
Endotoxin Analysis
LAL assay screening for bacterial endotoxins, kept well below industry safety thresholds.
ISO 17025 Compliance
Our laboratory operations are accredited under ISO 17025, the international standard for testing competence.
Molecular Specifications
———