Certificate of Analysis
Lab reports coming soon. HPLC, Mass Spec & endotoxin testing.
MSDS
Material Safety Data Sheet coming soon. Handling & storage info for laboratory use.
BPC-157
Arthro-Regenerative
Compound Overview
BPC-157, also known as Pentadecapeptide BPC-157, is a synthetic peptide composed of 15 amino acids derived from a naturally occurring gastric protein sequence. Within laboratory settings, BPC-157 has been studied in research involving tissue regeneration, angiogenesis, gastrointestinal integrity, inflammatory response pathways, and cellular repair processes. Its stability profile and broad investigational applications have made it one of the most recognised compounds within modern peptide research environments. Manufactured to strict research-grade standards, each batch undergoes independent full-panel analytical verification through ISO 17025 accredited laboratories for purity, identity, and consistency.
- Tissue regeneration research
- Gastrointestinal integrity studies
- Angiogenesis and vascular response pathways
- Cellular repair process research
- Tendon and connective tissue studies
- Inflammatory response pathway research
Each Synapse Lab batch undergoes:
- 99%+ purity verification
- Identity confirmation
- Full-panel analytical testing
- Batch traceability
- QR-coded COA access
Verified through independent ISO 17025 accredited laboratories.
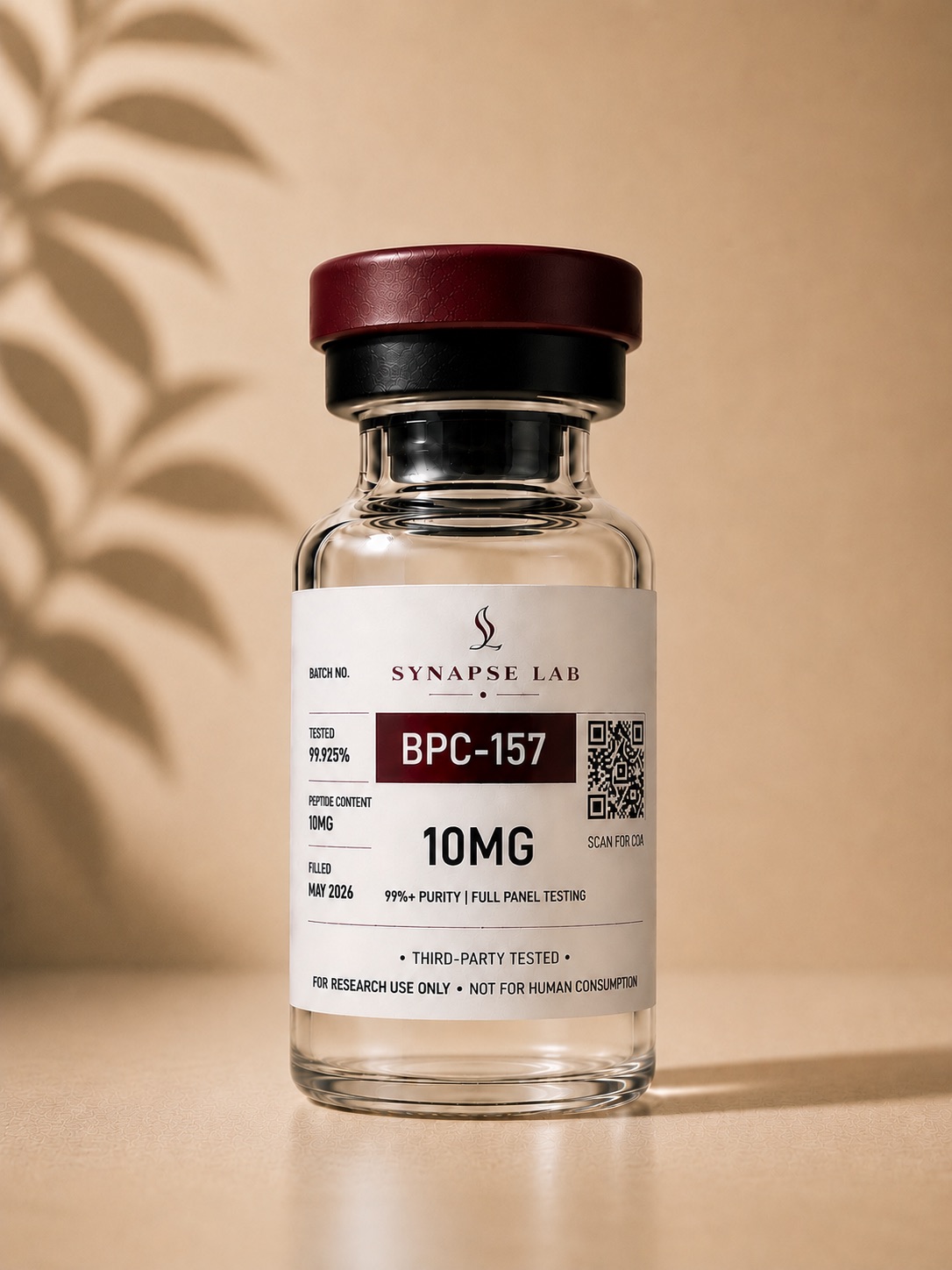
- Compound: BPC-157
- Peptide Type: Pentadecapeptide
- Purity: ≥99%
- Format: Lyophilized powder
- Fill Size: 10mg
- Storage: Refrigerated storage recommended
- Research Use Only
This compound is supplied strictly for laboratory and analytical research purposes only. Not for human consumption, therapeutic use, or diagnostic application.
The Synapse Quality Standard
Every compound undergoes a rigorous multi-stage verification process before receiving our seal of approval.
HPLC Testing
High-performance liquid chromatography ensures precise purity measurement for every synthesized batch.
Mass Spec Verification
Mass spectrometry confirms molecular identity and detects trace impurities at parts-per-billion sensitivity.
Endotoxin Analysis
LAL assay screening for bacterial endotoxins, kept well below industry safety thresholds.
ISO 17025 Compliance
Our laboratory operations are accredited under ISO 17025, the international standard for testing competence.
Molecular Specifications
Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val1419.5 g/mol137525-51-0


